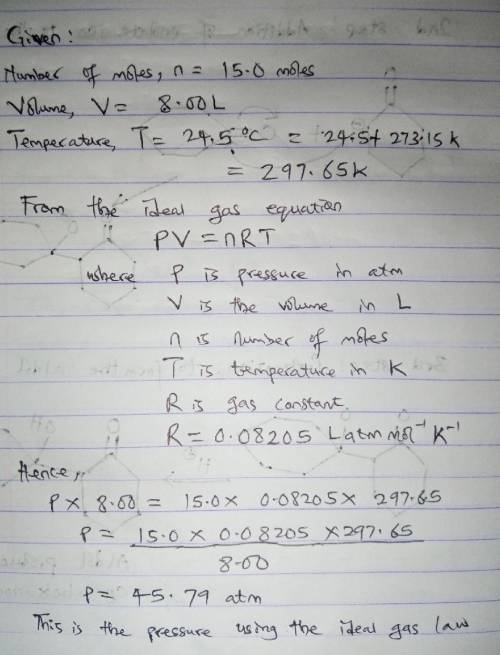Chemistry, 22.04.2020 02:08 awesomegrill
15.0 moles of gas are in a 8.00 LL tank at 24.5 ∘C∘C . Calculate the difference in pressure between methane and an ideal gas under these conditions. The van der Waals constants for methane are a=2.300L2⋅atm/mol2a=2.300L2⋅atm/mol 2 and b=0.0430 L/molb=0.0430 L/mol .

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2

Chemistry, 22.06.2019 22:00
Pls ill give u brainliest which of the following is true about science? 1. political conditions are unable to influence it. 2. economic concerns may prevent it from solving problems.
Answers: 2
You know the right answer?
15.0 moles of gas are in a 8.00 LL tank at 24.5 ∘C∘C . Calculate the difference in pressure between...
Questions


Mathematics, 04.09.2021 04:30



Mathematics, 04.09.2021 04:40


Physics, 04.09.2021 04:40


Mathematics, 04.09.2021 04:40

English, 04.09.2021 04:40





Mathematics, 04.09.2021 04:40


Mathematics, 04.09.2021 04:40

Business, 04.09.2021 04:40

Mathematics, 04.09.2021 04:40

Mathematics, 04.09.2021 04:40