
Chemistry, 22.04.2020 01:27 Queenvalentin
A 25.888 g sample of aqueous waste leaving a fertilizer manufacturer contains ammonia. The sample is diluted with 73.464 g of water. A 10.762 g aliquot of this solution is then titrated with 0.1039 M HCl . It required 31.89 mL of the HCl solution to reach the methyl red endpoint. Calculate the weight percent NH3 in the aqueous waste.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1
You know the right answer?
A 25.888 g sample of aqueous waste leaving a fertilizer manufacturer contains ammonia. The sample is...
Questions

Mathematics, 10.09.2020 07:01

English, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Biology, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01

Mathematics, 10.09.2020 07:01


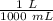
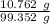
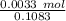
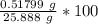 %
%

