
Chemistry, 21.04.2020 20:48 25jzaldivar
The decomposition reaction 2 NOCl → 2 NO + Cl_2 has a rate law that is second order with respect to [NOCl], where k = 3.2 M^{-1}s^{-1} at a certain temperature. If the initial concentration of NOCl is 0.076 M, how many seconds will it take for [NOCl] to decrease to 0.042 M at this temperature? Do not enter units with your numerical answer.

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 23.06.2019 03:00
In november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 1
You know the right answer?
The decomposition reaction 2 NOCl → 2 NO + Cl_2 has a rate law that is second order with respect to...
Questions

Mathematics, 24.04.2020 22:45


Biology, 24.04.2020 22:45


Chemistry, 24.04.2020 22:45

Social Studies, 24.04.2020 22:45

History, 24.04.2020 22:45










Mathematics, 24.04.2020 22:45

History, 24.04.2020 22:45


Physics, 24.04.2020 22:45

![\frac{1}{[NOCl]}=kt+\frac{1}{[NOCl]_{0}}](/tpl/images/0615/7124/81c05.png)
![[NOCl]_{0}](/tpl/images/0615/7124/069d3.png) is initial concentration of NOCl and k is rate constant.
is initial concentration of NOCl and k is rate constant.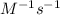 and [NOCl] = 0.042 M
and [NOCl] = 0.042 M![\frac{1}{0.042M}=[3.2M^{-1}s^{-1}\times t]+\frac{1}{0.076M}](/tpl/images/0615/7124/ee865.png)


