Chemistry, 21.04.2020 18:20 Isabella1319
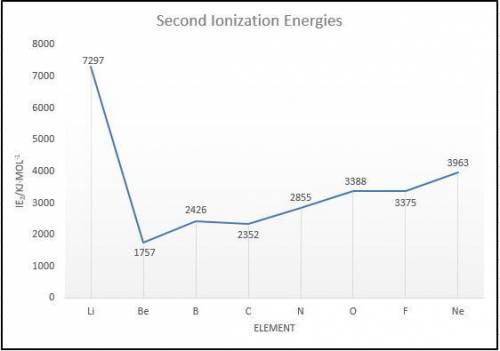
Elements in group 7A in the periodic table are called the halogens: elements in group 6A are called the chalcogens (a)
What is the most common oxidation state of the chalcogens compared to the halogens? (b) For each of the following
periodic properties. state whether the halogens or the chalcogens have larger values: atomic radii, ionic radii of the most
common oxidation state, first ionization energy. second ionization energy.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2
You know the right answer?
Elements in group 7A in the periodic table are called the halogens: elements in group 6A are called...
Questions





English, 02.09.2019 16:30


Computers and Technology, 02.09.2019 16:30

Biology, 02.09.2019 16:30

Social Studies, 02.09.2019 16:30

Mathematics, 02.09.2019 16:30

Mathematics, 02.09.2019 16:30



History, 02.09.2019 16:30

Spanish, 02.09.2019 16:30

History, 02.09.2019 16:30

Chemistry, 02.09.2019 16:30

History, 02.09.2019 16:30

Mathematics, 02.09.2019 16:30

English, 02.09.2019 16:30