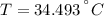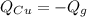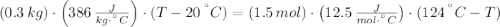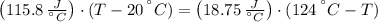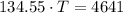A sealed copper container with a mass of 0.3 kg is filled with 1.5 mole of helium gas. Initially, the helium gas is at a temperature of 124 oC and the copper container is at 20 oC. The helium-copper system is thermally isolated. Note that the specific heat of copper is 386 J/(kgK) and the molar specific heat of helium is 12.5 J/(molK). Find the equilibrium temperature of the system.

Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:02
When undergoing chemical reactions where does the reactant combine to the enzyme?
Answers: 1

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2
You know the right answer?
A sealed copper container with a mass of 0.3 kg is filled with 1.5 mole of helium gas. Initially, th...
Questions



Social Studies, 19.09.2019 06:30

Mathematics, 19.09.2019 06:30

Social Studies, 19.09.2019 06:30

Mathematics, 19.09.2019 06:30

Mathematics, 19.09.2019 06:30

Biology, 19.09.2019 06:30

History, 19.09.2019 06:30





Biology, 19.09.2019 06:30



Chemistry, 19.09.2019 06:30