Chemistry, 20.04.2020 20:32 momneedshelphmwk
A 0.5781 g sample of a pure soluble bromide compound is dissolved in water, and all of the bromide ion is precipitated as AgBr by the addition of an excess of silver nitrate. The mass of the resulting AgBr is found to be 1.1166 g. What is the mass percentage of bromine in the original compound? %

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 14:50
How are evaporation and sublimation similar? a both involve the formation of a gas. b both release energy to the surroundings. c both take place throughout a solid. d both take place at the surface of a liquid.
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 22.06.2019 23:00
What is formed when amino acids form long chains or polymerize
Answers: 1
You know the right answer?
A 0.5781 g sample of a pure soluble bromide compound is dissolved in water, and all of the bromide i...
Questions

Mathematics, 10.04.2021 22:20



Health, 10.04.2021 22:30







Mathematics, 10.04.2021 22:30


Mathematics, 10.04.2021 22:30


Mathematics, 10.04.2021 22:30


English, 10.04.2021 22:30


Mathematics, 10.04.2021 22:30

Social Studies, 10.04.2021 22:30

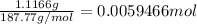
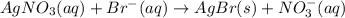
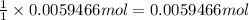 of bromide ions
of bromide ions