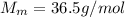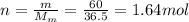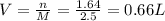Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Asample of silver (with work function ? = 4.52 ev) is exposed to an ultraviolet light source (? = 200 nm), which results in the ejection of photoelectrons. what changes will be observed if: silver is replaced with copper (? = 5.10 ev) more photoelectrons ejected no photoelectrons are emitted fewer photoelectrons ejected more energetic photoelectrons (on average) less energetic photoelectrons (on average)
Answers: 3

Chemistry, 22.06.2019 04:30
What are the primary responsibilities of a chemical engineer involved in "r& d"? develop large scale manufacturing operations discover new products and processes training of new chemists determine products needed by consumers
Answers: 2

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

You know the right answer?
60 g of HCL will make how many L of 2.5M HCL? (Molar Mass of HCL = 36.5g/mol)...
Questions



Chemistry, 22.04.2020 22:04









Social Studies, 22.04.2020 22:04



Biology, 22.04.2020 22:04


History, 22.04.2020 22:04

Mathematics, 22.04.2020 22:04

Biology, 22.04.2020 22:04

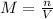 (1)
(1)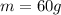 is the mass of the solute (HCl)
is the mass of the solute (HCl)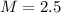 is the molarity of the solution
is the molarity of the solution