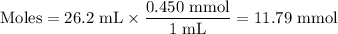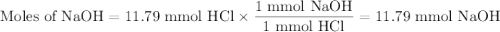Chemistry, 16.04.2020 03:18 Kingmoney959
26.2 mL of a 0.450 M hydrochloric acid solution is titrated with an unknown concentration of sodium hydroxide. 45.8 mL of the sodium hydroxide solution is required to reach the equivalence point. What is the molar concentration of the sodium hydroxide solution?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 02:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1


Chemistry, 23.06.2019 09:00
Need ! assume that the variables x and y are directly related. if k = 8, what is the value for each of the following points? be sure and record your data to be used in the following problem. x y k 0.
Answers: 2
You know the right answer?
26.2 mL of a 0.450 M hydrochloric acid solution is titrated with an unknown concentration of sodium...
Questions





Social Studies, 30.08.2019 13:30


History, 30.08.2019 13:30

English, 30.08.2019 13:30

Health, 30.08.2019 13:30




Spanish, 30.08.2019 13:30

Mathematics, 30.08.2019 13:30

Business, 30.08.2019 13:30


Mathematics, 30.08.2019 13:30