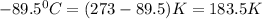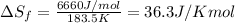Chemistry, 16.04.2020 02:53 tylijahking
Calculate the change in entropy that occurs in the system when 1.24 mol of isopropyl alcohol (c3h8o) melts at its melting point (-89.5 ∘c). heat of fusion is 5.37 kj/mol.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ineed this asap part i: scientific method what is the difference between science and pseudoscience? what is the scientific method?
Answers: 2

Chemistry, 22.06.2019 14:10
Precision can be defined as the o exact center of a data set. o reproducibility of a measured value. o correlation between two variables that are measured in a data set agreement between a measured value and an accepted value.
Answers: 2

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3
You know the right answer?
Calculate the change in entropy that occurs in the system when 1.24 mol of isopropyl alcohol (c3h8o)...
Questions







History, 15.04.2020 01:35









English, 15.04.2020 01:35

Computers and Technology, 15.04.2020 01:35

History, 15.04.2020 01:35


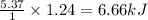
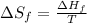
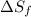 = Entropy of fusion = ?
= Entropy of fusion = ?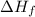 = enthalpy of fusion = 6.66 kJ/mol = 6660 J/mol (1kJ=1000J)
= enthalpy of fusion = 6.66 kJ/mol = 6660 J/mol (1kJ=1000J)