Chemistry, 16.04.2020 01:23 cschellfamily
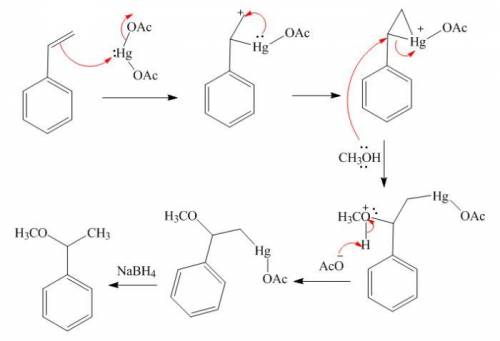
Styrene (1.53 g, 0.011 mol) in methanol (30 mL) was added to a mixture of Hg(OAc)2 (5.30 g, 0.016 mol) in methanol (100 mL) at room temp. and stirred for 24 h. Sodium hydroxide (3.0 M, 16 mL) was added, followed by NaBH4 (0.32 g, 0.008 mol) in NaOH (3.0 M, 16 mL) at 0 °C. The precipitated Hg was removed by filtration. The product was isolated by diethyl ether extraction. After drying over Na2SO4, solvent was removed and distillation gave the product. (Adapted from: Senda, Y.; Kanto, H.; Itoh, H. J. Chem. Soc., Perkin Trans. 2 1997, 1143-1146.)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 16:00
As changes in energy levels of electrons increase, the frequencies of atomic line spectra they emit
Answers: 2
You know the right answer?
Styrene (1.53 g, 0.011 mol) in methanol (30 mL) was added to a mixture of Hg(OAc)2 (5.30 g, 0.016 mo...
Questions



English, 06.05.2020 06:18


English, 06.05.2020 06:18

Health, 06.05.2020 06:18


Health, 06.05.2020 06:19


Mathematics, 06.05.2020 06:19


Mathematics, 06.05.2020 06:19

Spanish, 06.05.2020 06:19

Mathematics, 06.05.2020 06:19

Mathematics, 06.05.2020 06:19

English, 06.05.2020 06:19

Social Studies, 06.05.2020 06:19

Chemistry, 06.05.2020 06:19