

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 2

Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2
You know the right answer?
AB2 is a molecule that reacts readily with water. Calculate the bond energy of the A–B bond using th...
Questions

Physics, 01.12.2019 20:31

Biology, 01.12.2019 20:31

Chemistry, 01.12.2019 20:31


History, 01.12.2019 20:31


Social Studies, 01.12.2019 20:31




History, 01.12.2019 20:31








Computers and Technology, 01.12.2019 20:31

History, 01.12.2019 20:31

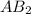 is a molecule that reacts readily with water. Calculate the bond energy of the A–B bond using the standard enthalpy of reaction and the bond energy data provided. Enter a number in kJ to 0 decimal places.
is a molecule that reacts readily with water. Calculate the bond energy of the A–B bond using the standard enthalpy of reaction and the bond energy data provided. Enter a number in kJ to 0 decimal places. 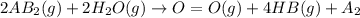
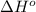 = –142 kJ
= –142 kJ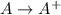
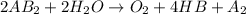
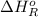 = -142 kJ
= -142 kJ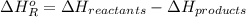
![[(2 \times 2 (A-B) + 2 \times 2 (O-H)] - [(O=O) + 4(H-B) + (A-A)]](/tpl/images/0603/6629/cfc5d.png)
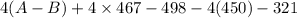
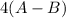 = -142 - 1868 + 498 + 1800 + 321
= -142 - 1868 + 498 + 1800 + 321

