Chemistry, 03.10.2019 10:30 andrewsaul04
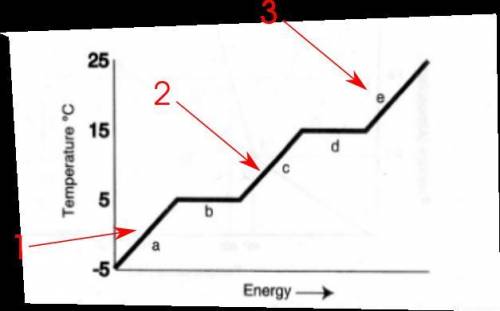
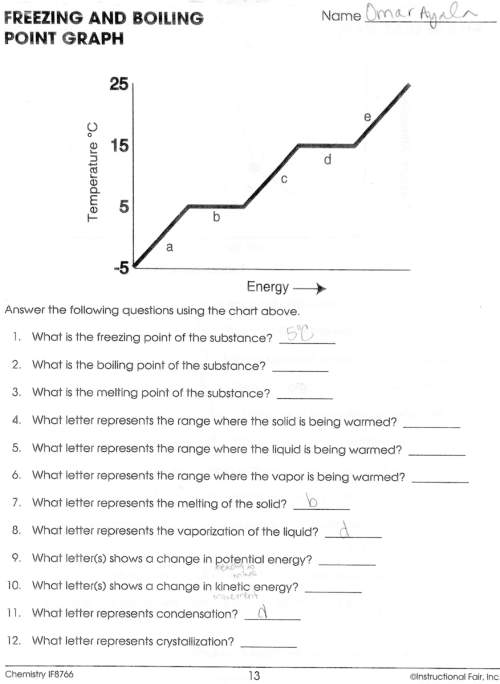
2. what is the boiling point of the substance?
3. what is the melting point of the substance?
4. what letter represents the range where the solid is being warmed?
5. what letter represents the range where the liquid is being warmed?
6. what letter represents the range where the vapor is being warmed?
9. what letters(s) shows a change in potential energy?
10. what letter(s) shows a change in kinetic energy?
and 12. what letter represents crystallization?
i've attached the file so you can see the freezing and boiling point graph.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 10:30
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1

Chemistry, 22.06.2019 18:50
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1

You know the right answer?
2. what is the boiling point of the substance?
3. what is the melting point of the substance?...
3. what is the melting point of the substance?...
Questions


Engineering, 23.01.2020 03:31

Mathematics, 23.01.2020 03:31


Mathematics, 23.01.2020 03:31

Mathematics, 23.01.2020 03:31

Mathematics, 23.01.2020 03:31

Mathematics, 23.01.2020 03:31



Mathematics, 23.01.2020 03:31



Biology, 23.01.2020 03:31

Mathematics, 23.01.2020 03:31


English, 23.01.2020 03:31

Mathematics, 23.01.2020 03:31


Mathematics, 23.01.2020 03:31