
Chemistry, 16.04.2020 00:50 jerenasmith77
What is the hydronium ion concentration of a 0.150 M hypochlorous acid solution with K a = 3.5 × 10-8? The equation for the dissociation of hypochlorous acid is:
HOCl(aq) + H2O(l) ⇌ H3O+(aq) + OCl-(aq).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
Un cierto gas tiene un volumen de 800ml a 80°c y 600ml a 80°c y 600mmhg de presión. ¿cual será el volumen del gas a condiciones normales? sí el gas es oxígeno, ¿cuál será su peso? y ¿cuántas moléculas están presentes en el sistema?
Answers: 2

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1
You know the right answer?
What is the hydronium ion concentration of a 0.150 M hypochlorous acid solution with K a = 3.5 × 10-...
Questions

Mathematics, 17.12.2020 22:20


Mathematics, 17.12.2020 22:20

Mathematics, 17.12.2020 22:20

Mathematics, 17.12.2020 22:20


Biology, 17.12.2020 22:20

Chemistry, 17.12.2020 22:20

Chemistry, 17.12.2020 22:20








Mathematics, 17.12.2020 22:20

Biology, 17.12.2020 22:20

Mathematics, 17.12.2020 22:20

![[H_3O^+]](/tpl/images/0603/9545/6cfd2.png) is
is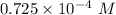 .
.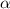 be change of concentration.
be change of concentration.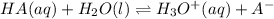
![K_a=\frac{[A^-][H_3O^+]}{[HA]}](/tpl/images/0603/9545/2421d.png)
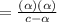
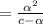
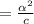 [ For weak acid , the value of
[ For weak acid , the value of 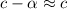 ]
]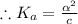
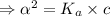
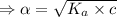
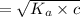
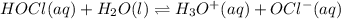
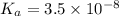
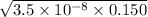 M
M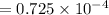 M
M

