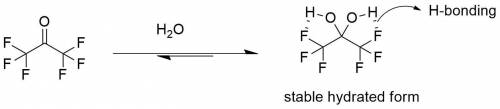
For most ketones, hydrate formation is unfavorable, because the equilibrium favors the ketone rather than the hydrate. However, the equilibrium for hydration of hexafluoroacetone favors formation of the hydrate. Provide a plausible explanation for this observation.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which describes interactions between substances and stomata during photosynthesis? check all that apply. oxygen enters stomata. oxygen is released through stomata. carbon dioxide enters stomata. carbon dioxide is released through stomata. hydrogen enters stomata. hydrogen is released through stomata.
Answers: 1

Chemistry, 22.06.2019 02:30
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1


Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2
You know the right answer?
For most ketones, hydrate formation is unfavorable, because the equilibrium favors the ketone rather...
Questions

Mathematics, 12.03.2020 16:11


English, 12.03.2020 16:12


Mathematics, 12.03.2020 16:12




Mathematics, 12.03.2020 16:12


Social Studies, 12.03.2020 16:12



English, 12.03.2020 16:13

Mathematics, 12.03.2020 16:13

Mathematics, 12.03.2020 16:13

Mathematics, 12.03.2020 16:13


Mathematics, 12.03.2020 16:13

Mathematics, 12.03.2020 16:13

 can not escape the hydrated form and goes back to keto form.
can not escape the hydrated form and goes back to keto form.