Chemistry, 15.04.2020 04:48 anitaabbey27
Linolenic acid (C18H30O2) can be hydrogenated to stearic acid by reacting it with hydrogen gas according to the equation: C18H30O2 + 3H2 --->C18H36O2 What volume of hydrogen gas, measured at STP, is required to react with 10.5 g of linolenic acid in this reaction?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Describing intermolecular forces use the drop down menus to match the type of intermolecular force to its name dipole dipole interactions dipole induced dipole interactions london dispersion forces hydrogen bond van der waals forces done
Answers: 1

Chemistry, 22.06.2019 15:30
The gulf stream is a warm water current that flows away from the equator to northern europe. witch of these does it cause. a. crashes of warm and cool water in the ocean b.colder climates near the equator c.large waves on the cost of europe d.warm climates in northern europe
Answers: 1

Chemistry, 22.06.2019 19:30
What is the common name for the compound shown here? enter the common name of the compound shown?
Answers: 2

Chemistry, 23.06.2019 03:10
Which is true according to the law of conservation of energy
Answers: 1
You know the right answer?
Linolenic acid (C18H30O2) can be hydrogenated to stearic acid by reacting it with hydrogen gas accor...
Questions


Social Studies, 04.11.2019 20:31

English, 04.11.2019 20:31

Mathematics, 04.11.2019 20:31

Mathematics, 04.11.2019 20:31



Mathematics, 04.11.2019 20:31


Mathematics, 04.11.2019 21:31

Mathematics, 04.11.2019 21:31




Mathematics, 04.11.2019 21:31





Mathematics, 04.11.2019 21:31

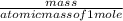
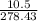
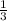 =
=