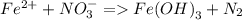Consider the following unbalanced redox reactions. In each case, separate the whole reactions into half-reactions, balance the half reactions, and combine to yield a balanced whole reaction. Compute the net reduction potential (E°) and pε° values for the net reactions and indicate whether the reaction is spontaneous as written. The reduction potential for Fe(OH)_3(s)/Fe^2+ is -0.181 V and pε is -3.08.
a. Fe^2 + + NO_3 = Fe(OH)_3(s) + N_2
b. Mn^2 + + Fe(OH)_3(s) = MnO_2(s) + Fe^2+

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 23.06.2019 03:00
In november 1987, a massive iceberg broke loose from the antartic ice mass and floated free in the ocean. the chunk of ice was estimated to be 98 mi long, 25 mi wide, and 750 ft thick. a typical backyard swimming pool contains about 24,000 gallons of water. how many of these pools could you fill from the water in this iceberg? (assume the iceberg is a rectangular solid of the above dimensions and consists of water only). express answer in scientific notation.
Answers: 3

Chemistry, 23.06.2019 14:00
[08.05] which answer provides the correct name for name the following hydrocarbon? (2 points) moving left to right: a hydrocarbon chain made of a methyl group (ch subscript three) single bond carbon triple bond carbon single bond ch bonded to a methyl group (ch subscript three) and a chain including a methelyne group (ch subscript two) single bond methyl group (ch subscript three). 4-ethyl-2-pentyne 4-methyl-2-hexyne 2-heptyne 4-methy-hexane
Answers: 1
You know the right answer?
Consider the following unbalanced redox reactions. In each case, separate the whole reactions into h...
Questions

Mathematics, 05.03.2020 08:41










Social Studies, 05.03.2020 08:42





Mathematics, 05.03.2020 08:43

Mathematics, 05.03.2020 08:43