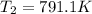Chemistry, 15.04.2020 02:57 christiannpettyy
The compound 1,1-difluoroethane decomposes at elevated temperatures to give fluoroethylene and hydrogen fluoride: CH3CHF2(g) → CH2CHF(g) + HF(g) At 460 °C, k = 5.8 × 10-6 s-1 and Ea = 265 kJ/mol. To what temperature (in K) would you have to raise the reaction to make it go four times as fast?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
The compound 1,1-difluoroethane decomposes at elevated temperatures to give fluoroethylene and hydro...
Questions


History, 20.08.2019 00:00

Mathematics, 20.08.2019 00:00


Mathematics, 20.08.2019 00:00

Mathematics, 20.08.2019 00:00

Mathematics, 20.08.2019 00:00

Physics, 20.08.2019 00:00

Mathematics, 20.08.2019 00:00

Geography, 20.08.2019 00:00



Geography, 20.08.2019 00:00


Mathematics, 20.08.2019 00:00




History, 20.08.2019 00:00

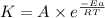
![\log (\frac{K_2}{K_1})=\frac{Ea}{2.303\times R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0600/9306/6d953.png)
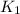 = rate constant at
= rate constant at 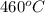 =
= 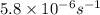
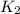 = rate constant at
= rate constant at 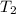 =
= 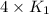
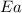 = activation energy for the reaction = 265 kJ/mol = 265000 J/mol
= activation energy for the reaction = 265 kJ/mol = 265000 J/mol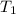 = initial temperature =
= initial temperature = 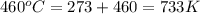
![\log (\frac{4\times K_1}{K_1})=\frac{265000J/mol}{2.303\times 8.314J/mole.K}[\frac{1}{733K}-\frac{1}{T_2}]](/tpl/images/0600/9306/ddf14.png)