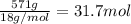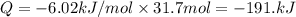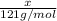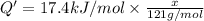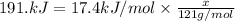A particular refrigerator cools by evaporating liquefied dichlorodifluoromethane, CCl2F2. How many kilograms of this liquid must be evaporated to freeze a tray of water at 0°C to ice at 0°C? The mass of the water is 571 g, the heat of fusion of ice is 6.02 kJ/mol, and the heat of vaporization of dichlorodifluoromethane is 17.4 kJ/mol.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Someone, part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 1

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2
You know the right answer?
A particular refrigerator cools by evaporating liquefied dichlorodifluoromethane, CCl2F2. How many k...
Questions

Mathematics, 01.02.2021 20:00


Mathematics, 01.02.2021 20:00

English, 01.02.2021 20:00

World Languages, 01.02.2021 20:00

Mathematics, 01.02.2021 20:00

English, 01.02.2021 20:00


English, 01.02.2021 20:00

Mathematics, 01.02.2021 20:00



Geography, 01.02.2021 20:00



Mathematics, 01.02.2021 20:00

Health, 01.02.2021 20:00


Mathematics, 01.02.2021 20:00

Chemistry, 01.02.2021 20:00