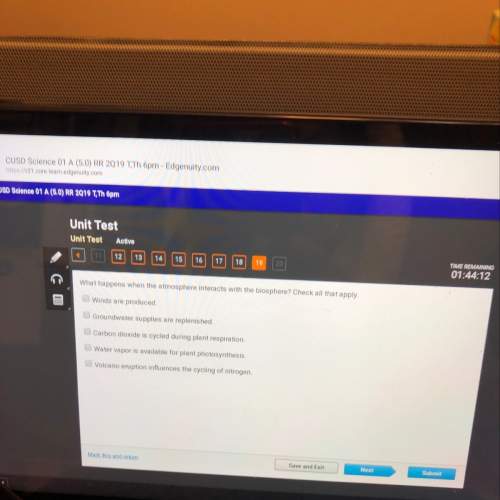
Which of the following statement(s) is/are consistent with the Bohr theory of the atom (and no other)?
(1) An electron can remain in a particular orbit as long as it continually absorbs radiation of a definite frequency.
(2) The lowest energy orbits are those closest to the nucleus.
(3) An electron can jump from the K shell (n = 1 major energy level) to the M shell (n = 3 major energy level) by absorbing radiation of a definite frequency.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:00
An example of technology is the a. addition of a side group to an organic molecule during synthesis. b. use of a new antibiotic to fight an infection. c. measurement of iron concentration in a water sample. d. study of atomic fusion reactions
Answers: 3

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1
You know the right answer?
Which of the following statement(s) is/are consistent with the Bohr theory of the atom (and no other...
Questions



Mathematics, 23.03.2020 21:46


Mathematics, 23.03.2020 21:46

Computers and Technology, 23.03.2020 21:46




Mathematics, 23.03.2020 21:46

Advanced Placement (AP), 23.03.2020 21:46

Mathematics, 23.03.2020 21:46

Mathematics, 23.03.2020 21:46

English, 23.03.2020 21:46

Mathematics, 23.03.2020 21:46

Biology, 23.03.2020 21:46




Mathematics, 23.03.2020 21:46