
Chemistry, 14.04.2020 16:12 buiratsamah
Butane (C4 H10(g), Hf = –125.6 kJ/mol) reacts with oxygen to produce carbon dioxide (CO2 , Hf = –393.5 kJ/mol ) and water (H2 O, Hf = –241.82 kJ/mol) according to the equation below. What is the enthalpy of combustion (per mole) of C4H10 (g)? Use .

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
How much heat is released upon converting one mole of steam (18.0 g) from 100.0 ∘c to water at 25.0 ∘c? show work and constants, trying to figure out how it works. only given the heat capacity for steam and water so try to only use that
Answers: 1


Chemistry, 22.06.2019 23:30
Why do oxygen have a strong attractive force for electrons
Answers: 2

Chemistry, 23.06.2019 00:30
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
You know the right answer?
Butane (C4 H10(g), Hf = –125.6 kJ/mol) reacts with oxygen to produce carbon dioxide (CO2 , Hf = –393...
Questions

Mathematics, 13.03.2021 14:00


History, 13.03.2021 14:00

Computers and Technology, 13.03.2021 14:00

English, 13.03.2021 14:00

Mathematics, 13.03.2021 14:00

Computers and Technology, 13.03.2021 14:00


English, 13.03.2021 14:00




Mathematics, 13.03.2021 14:00

Mathematics, 13.03.2021 14:00

English, 13.03.2021 14:00

English, 13.03.2021 14:00


History, 13.03.2021 14:00

Physics, 13.03.2021 14:00

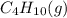 is -2657.5 kJ
is -2657.5 kJ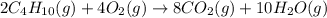
![\Delta H^o_{rxn}=[(8\times \Delta H^o_f_{CO_2(g)})+(10\times \Delta H^o_f_{H_2O(g)})]-[(1\times \Delta H^o_f_{C_4H_{10}(g)})+(4\times \Delta H^o_f_{O_2(g)})]](/tpl/images/0598/2199/66d33.png)
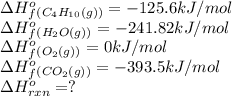
![\Delta H^o_{rxn}=[(8\times -393.5)+(10\times -241.82)]-[(2\times -125.6)+(4\times 0)]\\\\\Delta H^o_{rxn}=-5315kJ](/tpl/images/0598/2199/af1b6.png)


