
Chemistry, 08.04.2020 20:07 Darkknighta26
The reaction 2 n2o5(g) → 4 no2(g) + o2(g) is found to obey the rate law: rate = k [n2o5]. does the reaction mechanism consist of only a single bimolecular step? 1. yes, it must 2. it might, but you cannot tell from the information given. 3. no, it cannot

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
What is the volume occupied by 10.0 dm3 of gas at standard pressure after it has been compressedat constant temputure to 500.0 kpa?
Answers: 1

Chemistry, 22.06.2019 09:00
Plz mark brainliest 30 points1) find the momentum of a 12 kg snowball that is rolling with a velocity of 9 m/s.2) an 8 ball with a mass of .5 kg is sitting at rest. it is hit by the cue ball (1 kg) traveling at 2.5 m/s. if the cue ball is at rest after the collision, how fast is the 8 ball traveling after the collision? 3) two football players are running toward each other. if the offensive player is 75 kg and is running 8 m/s, how fast must the 60 kg defensive player run in order for the two players to hit and stop?
Answers: 1

Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1

Chemistry, 23.06.2019 03:00
Which of the following is a chemical property of water at 4 c
Answers: 2
You know the right answer?
The reaction 2 n2o5(g) → 4 no2(g) + o2(g) is found to obey the rate law: rate = k [n2o5]. does the r...
Questions

Social Studies, 23.12.2021 03:40

History, 23.12.2021 03:40


English, 23.12.2021 03:40



Computers and Technology, 23.12.2021 03:50

Mathematics, 23.12.2021 03:50

Mathematics, 23.12.2021 03:50









Computers and Technology, 23.12.2021 03:50


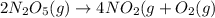
![Rate=k[N_2O_5]^1](/tpl/images/0589/8713/ae447.png)
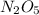 , it is a complex reaction.
, it is a complex reaction.

