
Chemistry, 08.04.2020 03:35 rainbowmc6
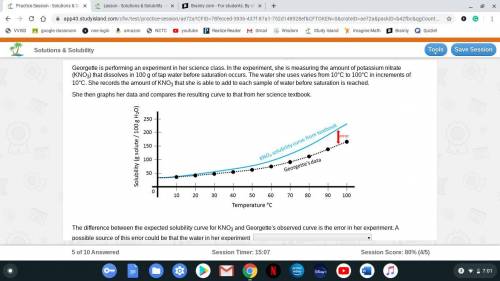
Georgette is performing an experiment in her science class. In the experiment, she is measuring the amount of potassium nitrate (KNO3) that dissolves in 100 g of tap water before saturation occurs. The water she uses varies from 10°C to 100°C in increments of 10°C. She records the amount of KNO3 that she is able to add to each sample of water before saturation is reached.
She then graphs her data and compares the resulting curve to that from her science textbook.
The difference between the expected solubility curve for KNO3 and Georgette's observed curve is the error in her experiment. A possible source of this error could be that the water in her experiment
A. did not have any substances already in it
B. already had substances dissolved in it
C. vaporized at about 40°C
D Solidified at about 40°C

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3


Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3
You know the right answer?
Georgette is performing an experiment in her science class. In the experiment, she is measuring the...
Questions

English, 13.11.2020 03:20

Mathematics, 13.11.2020 03:20


English, 13.11.2020 03:20



Biology, 13.11.2020 03:20

English, 13.11.2020 03:20

Mathematics, 13.11.2020 03:20

Social Studies, 13.11.2020 03:20

Mathematics, 13.11.2020 03:20



Mathematics, 13.11.2020 03:20

Computers and Technology, 13.11.2020 03:20

Social Studies, 13.11.2020 03:20




English, 13.11.2020 03:20



