
Chemistry, 08.04.2020 00:56 klmklm3799
Magnesium oxide can be made by heating magnesium metal in the presence of oxygen. The balanced equation for the reaction is: 2Mg(s)+O2(g)→2MgO(s) When 10.1 g of Mg are allowed to react with 10.5 g of O2, 10.3 g of MgO are collected. Dteremine the limiting reactants for the reaction

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:40
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na,so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1


Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2

You know the right answer?
Magnesium oxide can be made by heating magnesium metal in the presence of oxygen. The balanced equat...
Questions

Social Studies, 09.07.2019 16:00




Social Studies, 09.07.2019 16:00


History, 09.07.2019 16:00

Mathematics, 09.07.2019 16:00

Biology, 09.07.2019 16:00


Biology, 09.07.2019 16:00

Mathematics, 09.07.2019 16:00





Biology, 09.07.2019 16:00

Mathematics, 09.07.2019 16:00

Mathematics, 09.07.2019 16:00

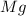 = 10.1 g
= 10.1 g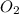 = 10.5 g
= 10.5 g = 32 g/mol
= 32 g/mol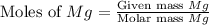
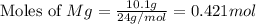
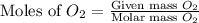
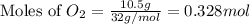
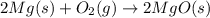
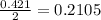 moles of
moles of 

