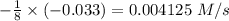Chemistry, 03.12.2019 16:31 madison1284
Consider the reaction: 8h2s(g)+4o2(g)→8h2o(g)+s8(g). δ[h2s]/δt = -0.033m/s. find δ[o2]/δt. δ[h2o]/δt. δ[s8]/δt. find the rate of the reaction.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Does anyone know a lot about how to: - calculate mass of magnesium metal - calculate the actual yield of magnesium oxide - calculate the theoretical yield of mgo - calculate the percent yield of mgo - determine the percent yield of mgo - determine the average percent yield of mgo i had to do an online lab and its asking these questions but i have no idea where to start or how to be able to find these things. i can post the chart of the data from the lab or if you can tell me exactly how i can find each.
Answers: 3

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1

You know the right answer?
Consider the reaction: 8h2s(g)+4o2(g)→8h2o(g)+s8(g). δ[h2s]/δt = -0.033m/s. find δ[o2]/δt. δ[h2o]/δ...
Questions



Social Studies, 24.03.2020 06:10















Computers and Technology, 24.03.2020 06:11


Computers and Technology, 24.03.2020 06:11

![\frac{[\Delta O_2]}{\Delta t} = -0.0165\ M](/tpl/images/0401/1414/d412a.png)
![\frac{[\Delta H_2O]}{\Delta t}= 0.033\ M/s](/tpl/images/0401/1414/ccd80.png)
![\frac{[\Delta S_8]}{\Delta t} = 0.004125\ M/s](/tpl/images/0401/1414/d55d1.png)
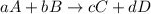
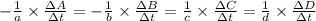
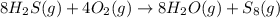
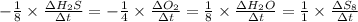
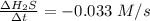
![-\frac{1}{8}\times \frac{[\Delta H_2S]}{\Delta t} =-\frac{1}{4}\times \frac{[\Delta O_2]}{\Delta t}](/tpl/images/0401/1414/fd6a9.png)
![-\frac{1}{8}\times (-0.33) =-\frac{1}{4}\times \frac{[\Delta O_2]}{\Delta t}](/tpl/images/0401/1414/02deb.png)
![-\frac{[\Delta O_2]}{\Delta t} = \frac{4}{8} \times (0.033) = 0.0165\ M](/tpl/images/0401/1414/323db.png)
![\frac{[\Delta O_2]}{\Delta t} = -0.0165\ M/s](/tpl/images/0401/1414/d516e.png)
![-\frac{1}{8}\times \frac{[\Delta H_2S]}{\Delta t} =\frac{1}{8}\times \frac{[\Delta H_2O]}{\Delta t}](/tpl/images/0401/1414/d13b5.png)
![\frac{[\Delta H_2O]}{\Delta t}=-\frac{8}{8}\times \frac{[\Delta H_2S]}{\Delta t}](/tpl/images/0401/1414/4e162.png)
![\frac{[\Delta H_2O]}{\Delta t}=-\frac{8}{8}\times (-0.033)](/tpl/images/0401/1414/b29cf.png)
![-\frac{1}{8}\times \frac{[\Delta H_2S]}{\Delta t} =\frac{1}{1}\times \frac{[\Delta S_8]}{\Delta t}](/tpl/images/0401/1414/1cad1.png)
![\frac{[\Delta S_8]}{\Delta t}=-\frac{1}{8}\times (-0.033)=0.004125\ M/s](/tpl/images/0401/1414/4b7e9.png)
![-\frac{1}{8}\times \frac{[\Delta H_2S]}{\Delta t}](/tpl/images/0401/1414/78436.png)