
Chemistry, 07.04.2020 22:38 JocelynC24
A certain amount of gas at 18.0°C and at a pressure of 0.900 atm is contained in a glass vessel. Suppose that the vessel can withstand a pressure of 1.90 atm. How high can you raise the temperature of the gas without bursting the vessel? In other words, at what temperature will the glass vessel shatter, in degrees Celsius.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:50
Which best describes why nh4+ can form an ionic bond with cl-?
Answers: 3

Chemistry, 22.06.2019 14:50
Given the following information: mass of proton = 1.00728 amu mass of neutron = 1.00866 amu mass of electron = 5.486 × 10^-4 amu speed of light = 2.9979 × 10^8 m/s calculate the nuclear binding energy (absolute value) of 3li^6. which has an atomic mass of 6.015126 amu. j/mol.
Answers: 2

Chemistry, 23.06.2019 01:00
Iron (fe) reacts with copper sulfate (cuso4) to form iron (ii) sulfate. in this reaction, cu2+ gains electrons to form cu. which statement is true about this reaction? fe(s) + cuso4(aq) → feso4(aq) + cu(s)
Answers: 3

Chemistry, 23.06.2019 07:00
Choose the correct statement about licensed veterinarians in the united states. a. they must be certified by the avma. b. they can treat all nonhuman animals. c. they can can treat only animals specified on the license. d. they must choose a specialty.
Answers: 2
You know the right answer?
A certain amount of gas at 18.0°C and at a pressure of 0.900 atm is contained in a glass vessel. Sup...
Questions

Mathematics, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00





Mathematics, 18.11.2020 01:00

History, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00







Mathematics, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00

Mathematics, 18.11.2020 01:00


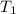 = 18 °c = 291 K
= 18 °c = 291 K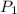 = 0.9 atm
= 0.9 atm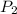 = 1.9 atm
= 1.9 atm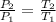
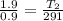
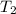 = 613.33 K = 341.33°c
= 613.33 K = 341.33°c

