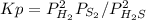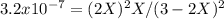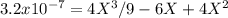Chemistry, 07.04.2020 19:18 litttyyyu33411
At 800 K, the equilibrium constant, Kp, for the following reaction is 3.2 × 10–7. 2 H2S(g) ⇌ 2 H2(g) + S2(g) A reaction vessel at 800 K initially contains 3.00 atm of H2S. If the reaction is allowed to equilibrate, what is the equilibrium pressure of S2?

Answers: 3


Another question on Chemistry


Chemistry, 23.06.2019 03:30
The molar mass of nickel(ni) is 58.7 g/mol. how many moles are in an 88 gram sample of nickel?
Answers: 1


Chemistry, 23.06.2019 04:31
What is the amount of energy for a photon that has a 125 cm wavelength
Answers: 2
You know the right answer?
At 800 K, the equilibrium constant, Kp, for the following reaction is 3.2 × 10–7. 2 H2S(g) ⇌ 2 H2(g)...
Questions



Mathematics, 25.10.2021 14:00




Mathematics, 25.10.2021 14:00

Mathematics, 25.10.2021 14:00


English, 25.10.2021 14:00


French, 25.10.2021 14:00




Biology, 25.10.2021 14:00

Mathematics, 25.10.2021 14:00