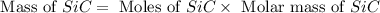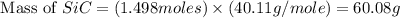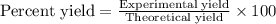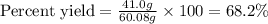Equation: SiO2 + 3C = SiC + 2CO When 90.0 g of silicon dioxide is heated with an excess of carbon, 41.0 g of silicon carbide is produced. What is the percent yield of this reaction? (find the theoretical amount of SiC using stoichiometry, then calculate percent yield)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 06:30
This drawing shows a human body system. what is the primary function of this body system?
Answers: 3

Chemistry, 22.06.2019 09:00
Ineed to find the answer of this question because i dont understand it
Answers: 1
You know the right answer?
Equation: SiO2 + 3C = SiC + 2CO When 90.0 g of silicon dioxide is heated with an excess of carbon, 4...
Questions

Mathematics, 30.06.2021 20:30

Mathematics, 30.06.2021 20:30



Mathematics, 30.06.2021 20:30








Mathematics, 30.06.2021 20:30






Mathematics, 30.06.2021 20:30

SAT, 30.06.2021 20:30

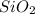 = 90.0 g
= 90.0 g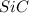 = 41.0 g
= 41.0 g
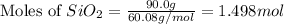
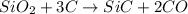
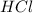 react to give 1.498 mole of
react to give 1.498 mole of