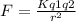Chemistry, 06.04.2020 03:29 7letters22
The first ionization energy of polonium, Po, is 812 kJ/mol. Is the first ionization energy of Lv expected to be greater than, less than, or equal to that of Po? Justify your answer in terms of Coulomb’s law.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 12:40
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1

Chemistry, 22.06.2019 23:30
The density of the solid phase of a substance is 0.90 g/cm3 and the density of the liquid phase is 1.0 g/cm3. a large increase in pressure will a. lower the freezing point b. raise the freezing point c. lower the boiling point d. raise the triple point e. lower the triple point
Answers: 1
You know the right answer?
The first ionization energy of polonium, Po, is 812 kJ/mol. Is the first ionization energy of Lv exp...
Questions

English, 02.12.2020 18:10


Biology, 02.12.2020 18:10

English, 02.12.2020 18:10


Chemistry, 02.12.2020 18:10

Health, 02.12.2020 18:10


Social Studies, 02.12.2020 18:10


Biology, 02.12.2020 18:10


Mathematics, 02.12.2020 18:10


History, 02.12.2020 18:10


English, 02.12.2020 18:10

History, 02.12.2020 18:10

Mathematics, 02.12.2020 18:10