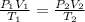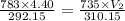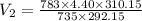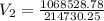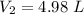During laparoscopic surgery, carbon dioxide gas is used to expand the abdomen to help create a larger working space.
If 4.40 L of CO2 gas at 19 ∘C at 783 mmHg is used, what is the final volume, in liters, of the gas at 37 ∘C and a pressure of 735 mmHg, if the amount of CO2 does not change?
Express your answer with the appropriate units.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 22:30
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1

Chemistry, 22.06.2019 23:00
What is the name of the enzyme that forms at the start of transcription?
Answers: 1

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1
You know the right answer?
During laparoscopic surgery, carbon dioxide gas is used to expand the abdomen to help create a large...
Questions

History, 20.03.2020 07:48

Computers and Technology, 20.03.2020 07:48

History, 20.03.2020 07:48


Mathematics, 20.03.2020 07:49


Mathematics, 20.03.2020 07:49

Mathematics, 20.03.2020 07:49










History, 20.03.2020 07:51

Mathematics, 20.03.2020 07:51

English, 20.03.2020 07:51