
Chemistry, 04.04.2020 11:01 deebroussard
Find the pH during the titration of 20.00 mL of 0.1000 M butanoic acid, CH3CH2CH2COOH (Ka = 1.54 x 10^-5), with 0.1000 M NaOH solution after the following additions of titrant.
(a) 10.00 mL: pH =
(b) 20.10 mL: pH =
(c) 25.00 mL: pH =

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 21.06.2019 17:50
An aqueous solution of potassium hydroxide is standardized by titration with a 0.194 m solution of hydrobromic acid. if 25.2 ml of base are required to neutralize 24.2 ml of the acid, what is the molarity of the potassium hydroxide solution? m potassium hydroxide
Answers: 2


Chemistry, 22.06.2019 04:00
14. many depressants reduce small muscle control, making it harder for a. you to steer b. your mind to consider complex problems c. the eye to scan, focus, or stay still d. the kidneys to filter alcohol out of the bloodstream
Answers: 3
You know the right answer?
Find the pH during the titration of 20.00 mL of 0.1000 M butanoic acid, CH3CH2CH2COOH (Ka = 1.54 x 1...
Questions





Mathematics, 13.04.2021 01:00



Mathematics, 13.04.2021 01:00





Mathematics, 13.04.2021 01:00



Mathematics, 13.04.2021 01:00

Chemistry, 13.04.2021 01:00

Mathematics, 13.04.2021 01:00


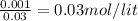
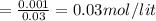
![pH=pK_{a}+log\frac{[conjugate base]}{[acid]} \\\\=-log(1.54X10^{-5} )+log\frac{0.03}{0.03} \\\\=4.81](/tpl/images/0582/2071/3377e.png)




