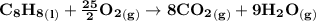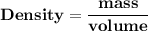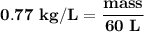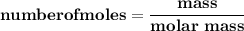Chemistry, 04.04.2020 07:07 selfiegymnast
How many L of carbon dioxide at 1.00 atm and 298.15 K are released from a car's engine upon consumption of a 60.0 L LIQUID tank gasoline? (Gasoline density: 0.77 kg/L, Molar mass of C₈H₁₈: 114.2 g/mol) C₈H₁₈ (l) + ²⁵/₂ O₂ (g) → 8 CO₂ (g) + 9 H₂O (g)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:30
One way that radioactive waste is treated is by burying it in repositories. the repositories are found only in states with very low populations. true or false? a. trueb. false(also i meant to put high school but it put down middle school instead)
Answers: 1

Chemistry, 22.06.2019 19:00
Which is the solubility product expression for caf2(s)?  [ca2+]/[f–]2  [ca2+][f2–]  [ca]+[f]2  [ca2+][f–]2
Answers: 3

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1

You know the right answer?
How many L of carbon dioxide at 1.00 atm and 298.15 K are released from a car's engine upon consumpt...
Questions

Mathematics, 07.01.2020 05:31







English, 07.01.2020 05:31


Physics, 07.01.2020 05:31


Physics, 07.01.2020 05:31


Advanced Placement (AP), 07.01.2020 05:31

History, 07.01.2020 05:31



Mathematics, 07.01.2020 05:31