Chemistry, 02.04.2020 18:45 wendymtz2004
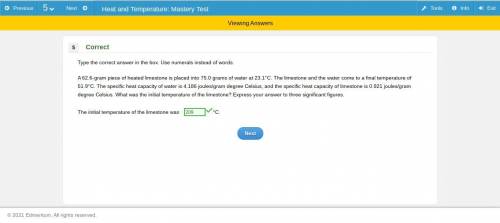
A 62.6-gram piece of heated limestone is placed into 75.0 grams of water at 23.1°C. The limestone and the water come to a final temperature of 51.9°C. The specific heat capacity of water is 4.186 joules/gram degree Celsius, and the specific heat capacity of limestone is 0.921 joules/gram degree Celsius. What was the initial temperature of the limestone? Express your answer to three significant figures

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 08:00
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1

Chemistry, 22.06.2019 12:00
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
You know the right answer?
A 62.6-gram piece of heated limestone is placed into 75.0 grams of water at 23.1°C. The limestone an...
Questions


Mathematics, 09.12.2019 06:31

History, 09.12.2019 06:31

Computers and Technology, 09.12.2019 06:31

Advanced Placement (AP), 09.12.2019 06:31


Mathematics, 09.12.2019 06:31

Mathematics, 09.12.2019 06:31

Mathematics, 09.12.2019 06:31


Mathematics, 09.12.2019 06:31

History, 09.12.2019 06:31

Advanced Placement (AP), 09.12.2019 06:31

Biology, 09.12.2019 06:31

History, 09.12.2019 06:31


Mathematics, 09.12.2019 06:31

English, 09.12.2019 06:31

Mathematics, 09.12.2019 06:31

Arts, 09.12.2019 06:31