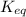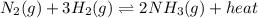Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 18:30
Which sample at stp has the same number of atoms as 18 liters of ne at stp
Answers: 1
You know the right answer?
Write the equilibrium expression for N2 (g) + 3 H2 (g) ↔ 2 NH3 (g) + heat I have no idea where to ev...
Questions




Mathematics, 07.08.2021 14:00

Mathematics, 07.08.2021 14:00

Social Studies, 07.08.2021 14:00

Mathematics, 07.08.2021 14:00

Computers and Technology, 07.08.2021 14:00

Arts, 07.08.2021 14:00


Health, 07.08.2021 14:00

English, 07.08.2021 14:00

Engineering, 07.08.2021 14:00

Biology, 07.08.2021 14:00

Mathematics, 07.08.2021 14:00

Mathematics, 07.08.2021 14:00




Health, 07.08.2021 14:00

![K_c=\frac{[NH_3]^2}{[N_2][H_2]^3}](/tpl/images/0576/2090/c3aa0.png)