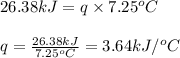Chemistry, 01.04.2020 01:20 hosteenimport21
What is the heat capacity of the calorimeter, Calorimeter, (in kJ/oC) of a bomb calorimeter if burning 1.0 g of benzoic acid in it causes the temperature to rise by 7.25 oC? Given ΔHcomb = LaTeX: -−26.38 kJ/g.
Answer Choices:
3221.53
3.64
191.26
25.34
-3.64

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:10
How does chemistry affect our world? a. chemicals makes our world more polluted. b. chemicals keeps us healthy. c. chemicals can or hurt our world. d. chemicals make our world safe to live in.
Answers: 1

Chemistry, 21.06.2019 18:00
During which movies do spring tides new moon first quarter waxing gibbous waxing
Answers: 1

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 12:00
A5.000 g sample of niso4 h2o decomposed to give 2.755 g of anhydrous niso4. what is the formula of the hydrate? what is the full chemical name for the hydrate? what is the molar mass of the hydrate? niso4•_h2o what is the mass % of water in the hydrate?
Answers: 1
You know the right answer?
What is the heat capacity of the calorimeter, Calorimeter, (in kJ/oC) of a bomb calorimeter if burni...
Questions





English, 16.09.2019 19:30

Computers and Technology, 16.09.2019 19:30

Computers and Technology, 16.09.2019 19:30


Biology, 16.09.2019 19:30



History, 16.09.2019 19:30


Social Studies, 16.09.2019 19:30

Mathematics, 16.09.2019 19:30

Chemistry, 16.09.2019 19:30

History, 16.09.2019 19:30

Chemistry, 16.09.2019 19:30


Mathematics, 16.09.2019 19:30

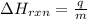
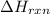 = enthalpy change of the reaction = -26.38 kJ/g
= enthalpy change of the reaction = -26.38 kJ/g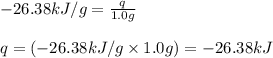
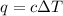
 = change in temperature = 7.25°C
= change in temperature = 7.25°C