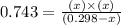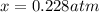Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1
You know the right answer?
At a certain temperature, the K p for the decomposition of H 2 S is 0.743 . H 2 S ( g ) − ⇀ ↽ − H 2...
Questions


History, 06.07.2019 13:00


Social Studies, 06.07.2019 13:00


Mathematics, 06.07.2019 13:00


Chemistry, 06.07.2019 13:00

Social Studies, 06.07.2019 13:00


Computers and Technology, 06.07.2019 13:00



Mathematics, 06.07.2019 13:00

Biology, 06.07.2019 13:00


Mathematics, 06.07.2019 13:00

Chemistry, 06.07.2019 13:00

Mathematics, 06.07.2019 13:00

Geography, 06.07.2019 13:00

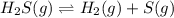
![K_{p}=\frac{[H_2]\times [S]}{[H_2S]}](/tpl/images/0572/1236/aa7b4.png)