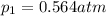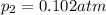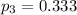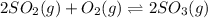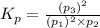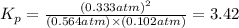Chemistry, 30.03.2020 19:17 animaljamissofab
The oxidation of sulfur dioxide by oxygen to sulfur trioxide has been implicated as an important step in the formation of acid rain: 2 SO2(g) + O2(g) ⇌ 2 SO3(g). If the equilibrium partial pressures of SO2, O2, and SO3 are 0.564 atm, 0.102 atm, and 0.333 atm respectively at 1000 K, what is K p at that temperature?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:00
How many mmols of tris-hcl are there in 100 ml of a 100 mm tris-hcl buffer solution at ph 8.1? note that the 100 mm refers to the sum of tris and tris-hcl concentrations?
Answers: 3

Chemistry, 22.06.2019 22:00
4.25g sample of solid ammonium nitrate dissolves in 60.0g of water in a coffee-cup calorimeter, the temperature drops from 22.0 c to 16.9 c. assume that the specific heat of the solution is the same as that of pure water. calculate delta(h) (in kj/mol nh4no3) for the solution proces.
Answers: 2

Chemistry, 23.06.2019 00:00
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
The oxidation of sulfur dioxide by oxygen to sulfur trioxide has been implicated as an important ste...
Questions

English, 24.02.2021 14:00


Business, 24.02.2021 14:00

Spanish, 24.02.2021 14:00



English, 24.02.2021 14:00


History, 24.02.2021 14:00

Mathematics, 24.02.2021 14:00



Mathematics, 24.02.2021 14:00





English, 24.02.2021 14:00

Biology, 24.02.2021 14:00

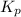 at this temperature is 3.42.
at this temperature is 3.42.