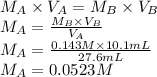Chemistry, 30.03.2020 19:38 cheaterman4121
A student is asked to standardize a solution of potassium hydroxide. He weighs out 1.08 g potassium hydrogen phthalate (KHC8H4O4, treat this as a monoprotic acid). It requires 36.8 mL of potassium hydroxide to reach the endpoint. A. What is the molarity of the potassium hydroxide solution? M This potassium hydroxide solution is then used to titrate an unknown solution of perchloric acid. B. If 10.1 mL of the potassium hydroxide solution is required to neutralize 27.6 mL of perchloric acid, what is the molarity of the perchloric acid solution? M

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2

Chemistry, 22.06.2019 10:00
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1


Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1
You know the right answer?
A student is asked to standardize a solution of potassium hydroxide. He weighs out 1.08 g potassium...
Questions

History, 30.07.2019 14:30


Biology, 30.07.2019 14:30

History, 30.07.2019 14:30


History, 30.07.2019 14:30

Mathematics, 30.07.2019 14:30







History, 30.07.2019 14:30

Business, 30.07.2019 14:30



History, 30.07.2019 14:30

Social Studies, 30.07.2019 14:30