Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Consider the nuclear equation below. 239 > x + 4 he 94 2 what is x? 1.235 cm 96 2.243 u 92 3.235 u 92 4.243 cm 96
Answers: 2

Chemistry, 21.06.2019 21:30
Which statements are true about electrolysis? check all that apply. electrolysis requires an acid be present. electrolysis is described by two half-reactions. electrolysis is not an industrial process. electrolysis results in commercially valuable products. electrolysis involves the transfer of electrons. reduction results in the loss of electrons. oxidation results in the loss of electrons.
Answers: 1

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3
You know the right answer?
In a liquid mixture of chloroform (C) and acetone (A) with chloroform mole fraction xC = 0.4693, the...
Questions

Mathematics, 17.03.2020 21:57

Mathematics, 17.03.2020 21:57



World Languages, 17.03.2020 21:57

History, 17.03.2020 21:57

Geography, 17.03.2020 21:57







Mathematics, 17.03.2020 21:57


Mathematics, 17.03.2020 21:57

Mathematics, 17.03.2020 21:57



Mathematics, 17.03.2020 21:57

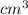
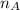
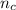
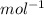
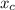 ) × 58.08 + n
) × 58.08 + n 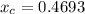
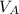 +
+