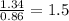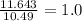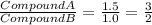Chemistry, 30.03.2020 16:16 kenishawilkinsoy4mgw
Sulfur reacts with oxygen and creates two compounds. Compound A contains 1.34 g of sulfur for every 0.86 g of oxygen. Compound B contains 11.63 g of sulfur for every 10.49 g of oxygen. What is the mass ratio of oxygen rounded to the nearest whole number

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:50
Write the overall equation for the reaction for lithium battery
Answers: 2

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2
You know the right answer?
Sulfur reacts with oxygen and creates two compounds. Compound A contains 1.34 g of sulfur for every...
Questions

Social Studies, 01.07.2021 15:20

Chemistry, 01.07.2021 15:30

Physics, 01.07.2021 15:30

Engineering, 01.07.2021 15:30

Social Studies, 01.07.2021 15:30

Mathematics, 01.07.2021 15:30


Mathematics, 01.07.2021 15:30

Advanced Placement (AP), 01.07.2021 15:30

English, 01.07.2021 15:30









Computers and Technology, 01.07.2021 15:30