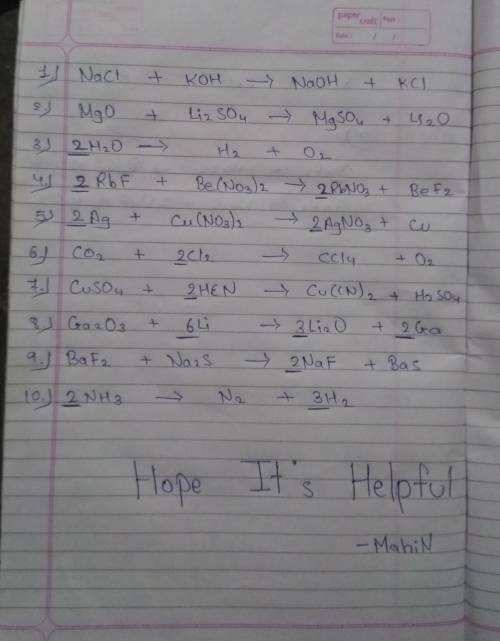1) NaCl + KOH NaOH + KCl
2) MgO + Li2SO4 MgSO4 + Li2O
3) H2O H...

Chemistry, 29.03.2020 19:13 christhegreat1
1) NaCl + KOH NaOH + KCl
2) MgO + Li2SO4 MgSO4 + Li2O
3) H2O H2 + O2
4) RbF + Be(NO3)2 RbNO3 + BeF2
5) Ag + Cu(NO3)2 AgNO3 + Cu
6) CO2 + Cl2 CCl4 + O2
7) CuSO4 + HCN Cu(CN)2 + H2SO4
8) Ga2O3 + Li Li2O + Ga
9) BaF2 + Na2S BaS2 + NaF
10) NH3 N2 + H2
please solve as many as you can, the boxes are arrows

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow.part ao2-(aq)+h2o(l)< => express your answer as part of a chemical equation. identify all of the phases in your answer.o2-(aq)+h2o(l) < => oh-(aq)+oh-(aq)part bpredict whether the equilibrium lies to the left or to the right of the equation in previous part.h2o is a stronger acid than oh–, so the equilibrium lies to the right.h2o is a weaker acid than oh–, so the equilibrium lies to the left.h2o is a stronger acid than oh–, so the equilibrium lies to the left.h2o is a weaker acid than oh–, so the equilibrium lies to the right.part cch3cooh(aq)+hs? (aq) < => express your answer as part of a chemical equation. identify all of the phases in your answer.ch3cooh(aq)+hs-(aq) < => h2s(aq)+c2h3o2-(aq)h2s(aq)+c2h3o2-(aq)part dpredict whether the equilibrium lies to the left or to the right of the equation in previous part.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the right.ch3cooh is a weaker acid than h2s, so the equilibrium lies to the left.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the right.ch3cooh is a stronger acid than h2s, so the equilibrium lies to the left.part eno2-(aq)+h2o(l) < => express your answer as part of a chemical equation. identify all of the phases in your answer.no2-(aq)+h2o(l) < => part fpredict whether the equilibrium lies to the left or to the right of the equation in previous part.hno2 is a stronger acid than h2o, so the equilibrium lies to the right.hno2 is a weaker acid than h2o, so the equilibrium lies to the left.hno2 is a stronger acid than h2o, so the equilibrium lies to the left.hno2 is a weaker acid than h2o, so the equilibrium lies to the right.
Answers: 1

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1

Chemistry, 22.06.2019 22:30
Calculate the concentration of all species in a 0.165 m solution of h2co3.
Answers: 1

Chemistry, 23.06.2019 06:00
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
You know the right answer?
Questions


Geography, 26.07.2019 04:30




Mathematics, 26.07.2019 04:30

English, 26.07.2019 04:30




SAT, 26.07.2019 04:30


Mathematics, 26.07.2019 04:30

Mathematics, 26.07.2019 04:30




History, 26.07.2019 04:30

Mathematics, 26.07.2019 04:30

Social Studies, 26.07.2019 04:30