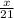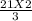Consider the following balanced reaction between hydrogen and nitrogen to form ammonia:
...

Chemistry, 28.03.2020 22:17 NetherisIsTheQueen
Consider the following balanced reaction between hydrogen and nitrogen to form ammonia:
3H2(g)+N2(g)→2NH3(g)
How many moles of NH3 can be produced from 21.0 mol of H2 and excess N2?

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 22:30
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1

Chemistry, 22.06.2019 13:20
Can someone me with 3 and 4 plz. this is for masteries test.
Answers: 2

Chemistry, 22.06.2019 21:00
Read "who built the pyramids? ”. leave this link open while you answer the questions throughout the assignment. give at least two reasons why some people claim the pyramids of giza were constructed by aliens.
Answers: 1
You know the right answer?
Questions





Mathematics, 17.10.2019 19:00

Social Studies, 17.10.2019 19:00



Biology, 17.10.2019 19:00

Biology, 17.10.2019 19:00


Mathematics, 17.10.2019 19:00

Social Studies, 17.10.2019 19:00


History, 17.10.2019 19:10


English, 17.10.2019 19:10

Mathematics, 17.10.2019 19:10


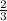 =
=