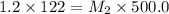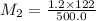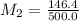Chemistry, 27.03.2020 22:58 neilaamusic6966
A 122mL sample of a 1.2 M sucrose solution is diluted to 500.0 mL. what is the molarity of the diluted solution

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 23.06.2019 06:00
Give one example of a pure (exact) number and of an estimated (measured) number.
Answers: 2
You know the right answer?
A 122mL sample of a 1.2 M sucrose solution is diluted to 500.0 mL. what is the molarity of the dilut...
Questions


Biology, 03.09.2019 19:10


Advanced Placement (AP), 03.09.2019 19:10



Physics, 03.09.2019 19:10


English, 03.09.2019 19:10

English, 03.09.2019 19:10

Social Studies, 03.09.2019 19:10





Computers and Technology, 03.09.2019 19:10