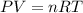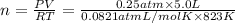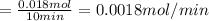Calcium oxide (CaO), an important ingredient in cement, is produced by decomposing calcium carbonate (CaCO3) at high temperature: CaCO3(s) → CaO(s) + CO2(g) In one particular reaction, 25 g of CaCO3 is heated at 550°C in a 5.0 L vessel. The pressure of CO2 is 0.25 atm after 10.0 minutes. What is the average rate of CO2 production in moles per minute during the 10 minutes? (Enter in mol/min.)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Which of the following statements concerning the influence of culture on ethnic identity formation is accurate? a. one will reject ethnic identity if cultural stereotypes are encountered. b. if one’s ethnic city is different from the dominant cultural group, then one’s ethnic identity you will become weekend. c. if an the ethnic group is excepted by dominant culture, then ethnic identity formation can be a difficult process. d. similarity to the dominant culture can determine how easy it is for one to except ethnic differences.
Answers: 2

Chemistry, 22.06.2019 08:30
How does the principle of electromagnetism explain the interaction between earth’s magnetic field and the solar wind?
Answers: 1

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2
You know the right answer?
Calcium oxide (CaO), an important ingredient in cement, is produced by decomposing calcium carbonate...
Questions

Engineering, 13.11.2020 03:20


Mathematics, 13.11.2020 03:20


Mathematics, 13.11.2020 03:20




Mathematics, 13.11.2020 03:20



Mathematics, 13.11.2020 03:20


Mathematics, 13.11.2020 03:20

Social Studies, 13.11.2020 03:20

Engineering, 13.11.2020 03:20


Mathematics, 13.11.2020 03:20

Computers and Technology, 13.11.2020 03:20