
Chemistry, 27.03.2020 01:29 mcckenziee
When the oxide of generic metal M is heated at 25.0 ∘ C , a negligible amount of M is produced. MO 2 ( s ) − ⇀ ↽ − M ( s ) + O 2 ( g ) Δ G ∘ = 291.0 kJ mol When this reaction is coupled to the conversion of graphite to carbon dioxide, it becomes spontaneous. What is the chemical equation of this coupled process? Show that the reaction is in equilibrium. Include physical states and represent graphite as C ( s ) .

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The variability in marine salinity between habitats does not impact the fish living there. select the best answer from the choices provided t f
Answers: 1

Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3

Chemistry, 22.06.2019 20:00
Glucose (c6h12o6) is an important biological molecule. (round the answer to nearest hundredth.) what is the percent by mass of carbon in glucose?
Answers: 2

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
When the oxide of generic metal M is heated at 25.0 ∘ C , a negligible amount of M is produced. MO 2...
Questions



English, 06.10.2019 02:30



History, 06.10.2019 02:30



Mathematics, 06.10.2019 02:30


Biology, 06.10.2019 02:30

Biology, 06.10.2019 02:30


Mathematics, 06.10.2019 02:30

Mathematics, 06.10.2019 02:30



Mathematics, 06.10.2019 02:30

Chemistry, 06.10.2019 02:30

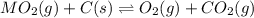
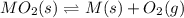 .....[1]
.....[1]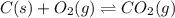 ..[2]
..[2]

