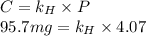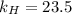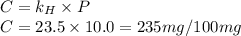Chemistry, 26.03.2020 22:40 xxgissellexx
At a certain temperature, the solubility of N2 gas in water at 4.07 atm is 95.7 mg of N2 gas/100 g water . Calculate the solubility of N2 gas in water, at the same temperature, if the partial pressure of N2 gas over the solution is increased from 4.07 atm to 10.0 atm .

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:30
Select the correct answer from each drop-down menu. daniel and sanya are scientists. daniel is studying whether the increasing frequency of tropical storms is affecting coastal erosion. sanya is investigating whether the discharge from industrial plants has any impact on the ph concentration of freshwater swamps in the surrounding area. which fields of science are daniel’s and sanya’s studies most closely related to? daniel’s field of study is related to science, and sanya’s field of study is related to .
Answers: 3

Chemistry, 21.06.2019 20:30
What was the procedure by which case united states vs lopez went to court
Answers: 1

Chemistry, 22.06.2019 00:00
Several kinds of bears are found on earth. most bears are brown or black, but one type of bear, the polar bear, is white. what process led to this difference in fur color? explain your answer.
Answers: 1

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3
You know the right answer?
At a certain temperature, the solubility of N2 gas in water at 4.07 atm is 95.7 mg of N2 gas/100 g w...
Questions

Mathematics, 19.11.2020 01:30


Social Studies, 19.11.2020 01:30

Mathematics, 19.11.2020 01:30

Mathematics, 19.11.2020 01:30



History, 19.11.2020 01:30




Mathematics, 19.11.2020 01:30

Mathematics, 19.11.2020 01:30

History, 19.11.2020 01:30

English, 19.11.2020 01:30

English, 19.11.2020 01:30



English, 19.11.2020 01:30

Chemistry, 19.11.2020 01:30

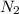 gas in water, at the same temperature, if the partial pressure of gas is 10.0 atm is 235mg/100g.
gas in water, at the same temperature, if the partial pressure of gas is 10.0 atm is 235mg/100g.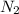 in water can be calculated by Henry’s Law. Henry’s law gives the relation between gas pressure and the concentration of dissolved gas.
in water can be calculated by Henry’s Law. Henry’s law gives the relation between gas pressure and the concentration of dissolved gas.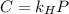 .
.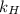 = Henry’s law constant = ?
= Henry’s law constant = ?