
Chemistry, 26.03.2020 20:48 rubycarbajal
The explosive nitroglycerin (C3H5N3O9) decomposes rapidly upon ignition or sudden impact according to the following balanced equation:
4C3H5N3O9(l)→12CO2(g)+10H2O(g)+6N2( g)+O2(g) ΔH∘rxn=−5678kJ
Required:
Calculate the standard enthalpy of formation (ΔH∘f) for nitroglycerin.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:50
How does a scientist the government? a. the scientist tells people in society what to do. b. the scientist determines the policies that the government spends money on. c. the scientist provides unbiased information to the government. d. the scientist makes laws based on his or her research results.
Answers: 1

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3

Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1
You know the right answer?
The explosive nitroglycerin (C3H5N3O9) decomposes rapidly upon ignition or sudden impact according t...
Questions

History, 16.04.2020 16:22



Chemistry, 16.04.2020 16:22



Mathematics, 16.04.2020 16:22





History, 16.04.2020 16:23


Mathematics, 16.04.2020 16:23

Computers and Technology, 16.04.2020 16:23





English, 16.04.2020 16:23

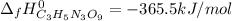
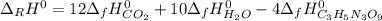
![\Delta _fH^0_{C_3H_5N_3O_9}=\frac{1}{4} (12\Delta _fH^0_{CO_2}+10\Delta _fH^0_{H_2O}-\Delta _RH^0)\\\Delta _fH^0_{C_3H_5N_3O_9}=\frac{1}{4mol} [12(-393.5kJ)+10(-241.8kJ)-(-5678kJ)]\\\Delta _fH^0_{C_3H_5N_3O_9}=-365.5kJ/mol](/tpl/images/0565/7176/fdf74.png)


