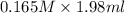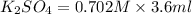Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Suppose you have designed a new thermometer called the x thermometer. on the x scale the boiling point of water is 129 ? x and the freezing point of water is 13 ? x. part a at what temperature are the readings on the fahrenheit and x thermometers the same?
Answers: 1

Chemistry, 22.06.2019 09:40
Apiece of copper has a temperature of 75.6 0c. when the metal is placed in 100.0 grams of water at 19.1 0c, the temperature rises by 5.5 0c. what is the mass of the metal?
Answers: 1


Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3
You know the right answer?
When aqueous solutions of K2SO4 and Pb(NO3)2 are combined, PbSO4 precipitates. Calculate the mass, i...
Questions

Mathematics, 26.04.2021 21:40



Mathematics, 26.04.2021 21:40


Social Studies, 26.04.2021 21:40

Chemistry, 26.04.2021 21:40

Spanish, 26.04.2021 21:40

Mathematics, 26.04.2021 21:40

Physics, 26.04.2021 21:40

English, 26.04.2021 21:40

English, 26.04.2021 21:40


English, 26.04.2021 21:40


Physics, 26.04.2021 21:40


Mathematics, 26.04.2021 21:40

History, 26.04.2021 21:40

Advanced Placement (AP), 26.04.2021 21:40

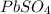 produced is 99.1 g.
produced is 99.1 g.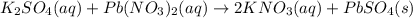
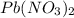 = Molarity × volume
= Molarity × volume