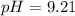Chemistry, 26.03.2020 17:49 seaotter9630
A chemist titrates 90.0 mL of a 0.5870 M acetic acid (HCH3CO2) solution with 0.4794M NaOH solution at 25 °C. Calculate the pH at equivalence. The p Kg of acetic acid is 4.76. Round your answer to 2 decimal places.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 07:30
Aradio signal from a gps satellite take only about 0.067 seconds to reach a gps reciever. if the speed of light is about 300,000km/s, then approximately how far away is the reciever from from the satellite?
Answers: 1

Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1
You know the right answer?
A chemist titrates 90.0 mL of a 0.5870 M acetic acid (HCH3CO2) solution with 0.4794M NaOH solution a...
Questions

Biology, 16.09.2021 01:10


Mathematics, 16.09.2021 01:10


Business, 16.09.2021 01:10

Mathematics, 16.09.2021 01:10

Computers and Technology, 16.09.2021 01:10


Chemistry, 16.09.2021 01:10






Mathematics, 16.09.2021 01:10


Mathematics, 16.09.2021 01:10



Computers and Technology, 16.09.2021 01:10

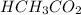 = 0.5870 M
= 0.5870 M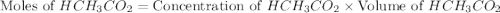
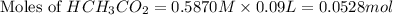
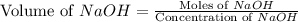
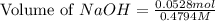
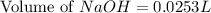
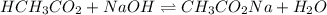
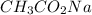 = 0.0528 mol
= 0.0528 mol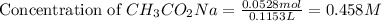
![pH=\frac{1}{2}[pK_w+pK_a+\log C]](/tpl/images/0565/3060/b44e5.png)
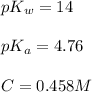
![pH=\frac{1}{2}[14+4.76+\log (0.458)]](/tpl/images/0565/3060/e66f0.png)