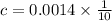Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 12:30
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1
You know the right answer?
What would be the composition and ph of an ideal buffer prepared from lactic acid (ch3chohco2h), whe...
Questions


Chemistry, 28.07.2019 19:50

Mathematics, 28.07.2019 19:50


Mathematics, 28.07.2019 19:50

English, 28.07.2019 19:50


History, 28.07.2019 19:50



Mathematics, 28.07.2019 19:50

Physics, 28.07.2019 19:50

Physics, 28.07.2019 19:50


Biology, 28.07.2019 19:50


Physics, 28.07.2019 19:50




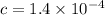
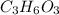 ⇄
⇄ 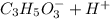
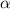 =1/10;
=1/10;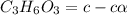
![[C_3H_5O_3^{-} ]= c\alpha](/tpl/images/0563/3127/9e63c.png)
![[H^{+}] = c\alpha](/tpl/images/0563/3127/f7069.png)
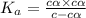
 can be neglected
can be neglected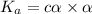
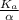
![P_H =- log[H^{+} ]](/tpl/images/0563/3127/1ad61.png)
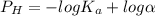
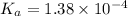
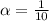
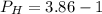
![c=\frac{1}{\alpha} \times [H^{+}]](/tpl/images/0563/3127/7fc04.png)
![[H^{+}] =antilog(-P_H)](/tpl/images/0563/3127/6a5c6.png)
![[H^{+} ] =0.0014](/tpl/images/0563/3127/a62ef.png)