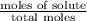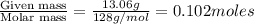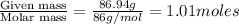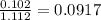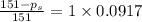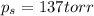Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 22:30
Consider a culture medium on which only gram-positive organisms such as staphylococcus aureus colonies can grow due to an elevated nacl level. a yellow halo surrounds the growth, indicating the bacterium fermented a sugar in the medium, decreasing the ph as a result and changing the color of a ph indicator chemical. this type of medium would be referred to as a differential and enrichment culture.
Answers: 2

Chemistry, 23.06.2019 06:00
Robert leaves a chocolate bar in his car while attending school all day. when he goes to his car in the afternoon, the bat has changed into gooey liquid. what happened to the chocolate bar
Answers: 1

Chemistry, 23.06.2019 08:00
The biosphere of the earth is made up of . a. inorganic b. organic
Answers: 2
You know the right answer?
A solution contains naphthalene (C10H8) dissolved in hexane (C6H14) at a concentration of 13.06% nap...
Questions


English, 08.02.2021 17:40



Computers and Technology, 08.02.2021 17:40


Mathematics, 08.02.2021 17:40

Social Studies, 08.02.2021 17:40


Mathematics, 08.02.2021 17:40

History, 08.02.2021 17:40

Mathematics, 08.02.2021 17:40

Mathematics, 08.02.2021 17:40

Mathematics, 08.02.2021 17:40

English, 08.02.2021 17:40



History, 08.02.2021 17:40

Mathematics, 08.02.2021 17:40

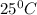 is 137 torr
is 137 torr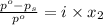
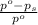 = relative lowering in vapor pressure
= relative lowering in vapor pressure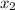 = mole fraction of solute =
= mole fraction of solute =